| RIKEN Center for
Developmental Biology (CDB) 2-2-3 Minatojima minamimachi, Chuo-ku, Kobe 650-0047, Japan |
| Head first: Novel method for the induction and regional specification of forebrain precursors from ES cells | ||
February 7, 2005 – The mammalian central nervous system is notoriously non-regenerative, and the treatment of disorders in which neural function has been compromised represents one of the greatest challenges remaining to modern medicine. Much attention has been paid in recent years to the promise of cell replacement therapy as a potential means to restore damaged nervous systems to health. This method, which involves the selective culture of cells for transplantation into patients deficient in a specific cell type, represents one of the best hopes on the horizon for patients suffering from a host of currently incurable afflictions.
This is the latest in a series of methodological advances for the high-efficiency differentiation of neurons from ES cells by the Sasai laboratory. In previous reports, Sasai described the application of a method based on the ability of feeder cells (called stromal cell derived inducing activity, or SDIA) used in combination with various growth factors to steer mouse and primate ES cells to differentiate into dopaminergic, sensory and enteric neurons at efficiencies of up to 90%. But these SDIA-based approaches failed to generate forebrain precursors (as distinguished by the expression of certain genes) at high frequency.
|
||
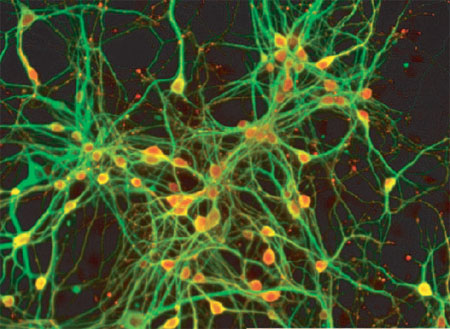 |
||
GABAergic basal telencephalic neurons (red) induced using the SFEB method |
||
The ability to generate the cellular forebears of the telencephalon from mouse ES cells represents a landmark in stem cell biology research. From a purely developmental perspective, the close mirroring of in vivo gene expression patterns by the externally induced effects seen in SFEB culture experiments provides new food for thought for scientists studying the genetic regulation of neurogenesis, particularly in its implications for the neural default model, which states that ectodermal cells tend to assume a neural fate in the absence of molecular messages to the contrary. And the proof-of-principle demonstration of the amenability of ES cells to forebrain differentiation and regional specification provides new avenues for biomedical researchers and clinicians to explore in the struggle to find cures for a range of human neurological disorders, including Huntington’s and Alzheimer’s disease, that affect the mind’s highest functions. |
||
|
||
[ Contact ] Douglas Sipp : sipp@cdb.riken.jp TEL : +81-78-306-3043 RIKEN CDB, Office for Science Communications and International Affairs |
| Copyright (C) CENTER FOR DEVELOPMENTAL BIOLOGY All rights reserved. |
