| RIKEN Center for Developmental Biology (CDB) 2-2-3 Minatojima minamimachi, Chuo-ku, Kobe 650-0047, Japan |
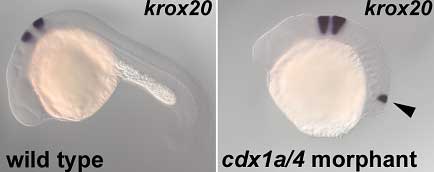
Now, in an article published in the journal Development, Takashi Shimizu and colleagues in the Laboratory for Vertebrate Axis Formation (Masahiko Hibi; Team Leader) reveal that a pair of Cdx-family proteins, Cdx1a and Cdx4, controls the neural tissue’s responsiveness to FGF and RA signaling. Studying the effects of the inhibition of these two factors in the embryonic zebrafish, they found that loss of Cdx1a/4 function induces the ectopic expression of markers of posterior hindbrain and anterior spinal cord, dependent on RA and FGF.
The team turned next to the FGFs and retinoic acid, which are also known to impact on hindbrain development, and whose opposing patterns of expression overlap at the approximate intersection of hindbrain and spinal cord. In the cdx morphants, these countervailing gradients also manifested in the caudal end, but were flipped along the A-P axis, with subtly different peaks of activity in cdx4 and cdx1a/4 embryos, with the region of maximal FGF and RA activity being more posterior in the double morphant. Suspecting that the region of overlapping FGF and RA signaling represented an ectopic environment that mimics the normal developmental field for the posterior hindbrain, Shimizu used specific inhibitors to interfere with FGF (fgf3 and fgf8) and retinoic acid signaling in the cdx morphants. While the perturbation of either of these signaling pathways had no discernible effect on the other, their loss of function did affect the expression of other hindbrain markers in the ectopic neural tissue, with FGFs responsible for markers for rhombomeres 4 to 6, and retinoic acid necessary for the ectopic expression of genes specific to the anterior spinal cord and the caudal-most region of the hindbrain. In cdx1a/4 morphant embryos in which both FGF and RA signaling was blocked, the mirror-image ectopic region failed to form. Of the genes whose expression was lost in the cdx1a/4 morphants, several were posterior-expressed members of the Hox family of patterning genes, suggesting that Hox signaling functions downstream of Cdx. Hox genes are known to work both as activators and repressors, prompting the team to try to untangle their role in this context. On constructing and expressing sequences in which either a repressor or an activator domain of was fused to a posterior Hox gene, the Hibi team found that while the repressor construct had no effect on ectopic gene expression in the cdx1a/4 morphant, the activator fusion protein inhibited both normal and ectopic expression of its target gene (as wild-type did), suggesting that the posterior Hox proteins function as transcriptional activators that indirectly suppress posterior hindbrain development. Shimizu’s findings highlight a surprisingly complex web of signaling at work in the formation of the posterior hindbrain and anterior spinal cord, in which Cdx activity in setting up regional competence to respond to FGF and retinoic acid signaling appears to be mediated by posterior members of the Hox family. In normal development, the expression of Cdx1a and -4 appear to control the responsiveness of the neural tissue to FGF and RA signals and suppress more anterior fates, such as hindbrain and anterior spinal cord; in the absence of inhibitory input from these Cdx factors, anterior tissues form ectopically. “Cdx genes were thought to control posterior body formation,” notes Hibi on the surprising findings, “but our study reveals that Cdx genes also function to repress anterior fate by causing a switch from tissue responsiveness to inductive signals.” |
|||||
|
|||||
 |
| Copyright (C) CENTER FOR DEVELOPMENTAL BIOLOGY All rights reserved. |