| RIKEN Center for Developmental Biology (CDB) 2-2-3 Minatojima minamimachi, Chuo-ku, Kobe 650-0047, Japan |
July 14, 2008 – Wnt activate pathways that play central roles in a wide range of developmental processes, from body axis specification to stem cell proliferation to morphogenetic cell movements. Two of the main programs are the canonical pathway, in which Wnt stabilizes ß-catenin allowing it to interact with various transcription factors in the nucleus, and the non-canonical, planar cell polarity (PCP) pathway, which features the activation of numerous small GTPases and downstream factors important in cell morphology and motility. Different Wnt ligands are associated with each of these pathways (Wnt 1, 3a and 8 are known as canonical, while Wnt5a and 11 are non-canonical), but the means by which pathways are selectively activated have remained unclear.
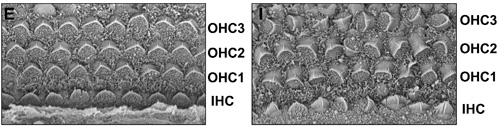
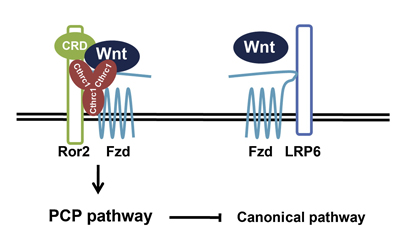
New work published by Shinji Yamamoto and others in the Laboratory for Embryonic Induction (Hiroshi Sasaki; Team Leader) shows for the first time how an extracellular cofactor enables the selective activation of a Wnt pathway. The report, published in Developmental Cell, reveals how a secreted glycoprotein, Cthrc1, helps switch on the PCP molecular cascade by stabilizing the interaction between Wnt ligands and their receptors. In the PCP pathway (as in canonical Wnt signaling) the primary receptor is the surface protein Frizzled, but importantly, the co-receptors in the two pathways differ; canonical signaling uses LRP5 and LRP6, while PCP uses a molecule named Ror2. Yamamoto’s first inkling of a role for Cthrc1 came from a screen for genes expressed in the notochord (a transient axial structure found in all chordate embryos). Drawn by the fact that it codes for a secreted protein expressed in both the notochord and inner ear, suggesting the possible involvement of the Wnt/PCP pathway, he first created a null mutant, but found no phenotype when Cthrc1 alone was ablated. When he studied its loss of function in mice carrying a heterozygous mutation in another PCP pathway element (Vangl2), however, he found defects in several developmental processes including neural tube closure and orientation of the cochlear hair cells in the inner ear.
Using co-culture immunoprecipitation, a method newly developed by the Sasaki lab to detect interactions between proteins, Yamamoto et al. next asked whether Cthrc1 binds with the co-receptor Ror2. Intriguingly, they found that it not only binds this PCP-specific co-receptor, but to Wnt ligands and the Frizzled receptor as well, opening up the possibility that Cthrc1 strengthens the interaction between components some of which are general to Wnt signaling and others that are specific to the PCP pathway. Conversely, Cthrc1 appears to suppress canonical Wnt signaling by some unknown mechanism. “The fact that the Cthrc1 solo knockout had no phenotype suggests that there are other genes able to compensate for its loss of function,” says Sasaki, “so one of the next challenges in this field will be to identify other genes that work in the selective activation of either the canonical or non-canonical Wnt pathways.”
|
|||||||
|
|||||||
|
|
| Copyright (C) CENTER FOR DEVELOPMENTAL BIOLOGY All rights reserved. |