| RIKEN Center for Developmental Biology (CDB) 2-2-3 Minatojima minamimachi, Chuo-ku, Kobe 650-0047, Japan |
August 5, 2008 – The hypothalamus is a small ventral region of the brain that sits atop the pituitary gland and serves as a critical interface between the nervous and endocrine systems. Although it only accounts for about 1/300 of the total brain mass, the hypothalamus plays important roles in the regulation of the autonomic nervous system and a set of behaviors that include feeding, sexuality, and the fight-or-flight response. Despite its importance, however, the development of the hypothalamus remains imperfectly understood, due in part to a lack of effective means for studying its differentiation in vitro. Now, new work by Takafumi Wataya and colleagues in the Laboratory for Organogenesis and Neurogenesis (Yoshiki Sasai; Group Director) demonstrating a technique for inducing hypothalamic differentiation from mouse embryonic stem (ES) cells in culture stands to add significantly to our understanding of the development of this tiny but complex brain region. In an article published in the Proceedings of the National Academy of Sciences USA, Wataya showed that, by modifying a technique used for inducing neuronal differentiation in pluripotent cells, it is possible to generate populations of cells expressing specific hypothalamic markers at high efficiency.
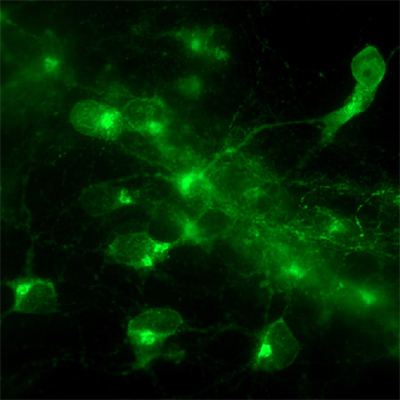
The study began when scientists in the Sasai lab noticed that mouse ES cells cultured using a method known as SFEB (for serum-free culture of embryoid body-like aggregates) very rarely expressed hypothalamic marker genes, although this technique had been used with great success in giving rise to other specific neuronal populations, including telencephalic (forebrain) precursors. “We knew that the medium typically used in SFEB culture contained a number of bioactive factors, such as insulin,” says Wataya, “so we tested whether these might somehow be interfering with hypothalamic differentiation.” Assays in which ES cells were SFEB-cultured in the presence or absence of insulin at different stages revealed that this hormone had a strong inhibitory effect on the differentiation of precursor cells expressing the hypothalamic marker gene, Rax, via the Akt pathway downstream. Interestingly, SFEB culture in the absence of insulin and other exogenous growth factors yielded populations of cells in which 60 to 70% expressed Rax and other genes indicative of hypothalamic neuronal fate. When the Rax-positive precursors were treated with the growth factor Sonic hedgehog, they found that cells went on to differentiate into neurons specific to different hypothalamic nuclei involved in hunger and satiety, which are of great potential value for studies of the cellular bases of metabolic regulation. “Hypothalamus function is implicated in a wide range of medical conditions, including appetite and metabolic problems such as anorexia and over-eating, but its small size has made it difficult to study in vivo,” says Wataya. “So we’re hopeful that the ability to grow large populations of homogeneous hypothalamic neurons through ES cell culture will make a contribution to the study of pathogenesis as well as the screening of new drug candidates.” The discovery that exogenous factors work to inhibit hypothalamic differentiation also intriguingly suggests that this rostral-ventral region may be thought of as a kind of zero-point in the Cartesian positional patterning of the early neuroectoderm. Taken together with the enormous biological importance and high degree of evolutionary conservation of this region, Wataya’s findings raise the interesting possibility that the hypothalamic precursor fate stands as a default outcome in brain development, opening new avenues of understanding of the evolution of nervous systems as well.
|
|||||
|
|||||
|
|
| Copyright (C) CENTER FOR DEVELOPMENTAL BIOLOGY All rights reserved. |