| RIKEN Center for Developmental Biology (CDB) 2-2-3 Minatojima minamimachi, Chuo-ku, Kobe 650-0047, Japan |
February 13, 2009 A new imaging technique developed through a collaboration between the CDB Laboratory for Stem Cell Biology (Shin-Ichi Nishikawa; Group Director) and Timm Schroeder, a former CDB research scientist who now heads the Independent Research Group for Hematopoiesis at the Helmholtz Zentrum München, German Research Center for Environmental Health, Institute for Stem Cell Research, has yielded a compelling new insights into the origins of blood in the mammalian embryo. This represents the first joint publication between the two institutes, which established a research affiliation in 2005.
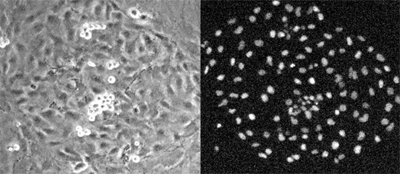
The debate over the ultimate source of embryonic hematopoiesis is fraught and longstanding, with competing hypotheses proposing that blood is generated from mesoderm, mesenchymal progenitors, endothelial precursors or cells in the endothelium itself. But limits in the technologies used to visualize blood differentiation at the cellular level by observing only single genetic markers at single points in time have meant that the existence of hemogenic endothelial cells has remained in dispute. Schroeder set out to address these shortcomings by developing a bio-imaging approach to enable him to watch well-characterized individual endothelial cells continuously during the differentiation process. Starting with mesodermal precursors (the cells from which endothelial cells originate) cultured under conditions known to induce endothelium, the team observed their morphology and behavior using computer-assisted time-lapse microscopy. From the total population, endothelial cells were isolated by selecting for the full set of their known characteristics, including sheet morphology, the ability to take up low-density lipoprotein, and the expression of VE-cadherin and the tight junction marker, claudin. Similarly, blood cells were identified by their non-adherence, proliferation, expression of blood-specific marker proteins, and the absence of endothelial characteristics. Looking at more than 10,000 mesodermal colonies, the team finally narrowed their focus down to 144 that had endothelial sheet morphology and generated blood cells. In all of these, they found that adherent endothelial cells gave rise to blood cells that gradually detached completely from the colony. The free-floating blood cells expressed blood lineage markers in a similar sequence to that of emerging hematopoietic cells in vivo. Importantly, the hemogenic endothelial cells failed to differentiate into either smooth muscle or cardiomyocytes, suggesting that they were not multipotent mesodermal cells. And they lacked the expression of CD45, a blood marker, further ruling out the possibility that they were bipotent blood-endothelial precursors. Indeed, the hemogenic cells gave rise solely to blood cells, while their neighboring sister cells generated only endothelial progeny. Mesodermal cells taken from mouse embryos showed similar hemogenic capacity, indicating that the findings were not merely an artifact of cell culture. |
||||||
|
||||||
 |
| Copyright (C) CENTER FOR DEVELOPMENTAL BIOLOGY All rights reserved. |