| RIKEN Center for Developmental Biology (CDB) 2-2-3 Minatojima minamimachi, Chuo-ku, Kobe 650-0047, Japan |
March 30, 2009 In the first few days of mammalian development, the embryo transforms from a clump of undifferentiated cells to a fluid-filled sphere, called the blastocyst, with distinct inner and outer cell types. The interior cells, known as the inner cell mass (ICM), go on to form the embryo proper, while the external layer of cells, or trophectoderm, contributes to extraembryonic tissues. Previous work has shown that the ICM-trophectoderm balance is maintained by interdependent mutual inhibition between a pair of factors: Cdx2, which drives trophectodermal differentiation, and Oct3/4, which maintains the ICM in an undifferentiated state. But the mechanisms involved in tipping this balance to either side in even earlier embryonic regions have never been worked out.
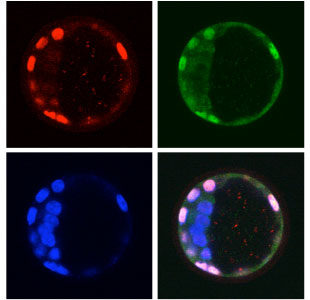
A new study by Noriyuki Nishioka and colleagues in the Laboratory for Embryonic Induction (Hiroshi Sasaki; Team Leader) now reveals that Hippo signaling controls the expression of both Cdx2 and Oct3/4 in the early embryo. The report, published in Developmental Cell, describes an intricate interplay between factors in the embryos inner and outer cells, in which the activation of the transcription factor Tead4 in outer cells switches on Cdx2 expression, while its suppression by the Hippo pathway in interior cells allows Oct3/4 to dominate. The authors speculate that this may be triggered by differences in the cell adhesion environments in these two zones of the embryo. Prior to the formation of the blastocyst, the cells in the embryo can only be distinguished from one another by their position in the exterior (surface) or interior of the embryo. It has been thought that positional information from the inner and/or outer cells might influence the contest between Cdx2 and Oct3/4 that defines the initial differentiation events in the blastocyst, but the mechanism to account for this remained elusive. There was, however, one tantalizing clue from previous work by the lab, which had showed that Tead4 is required for Cdx2 expression and the formation of the trophectoderm. The mystery was that Tead4 is expressed in every cell of the embryo, not just the cells that differentiate into trophectoderm. Another member of the Sasaki team, Mitsunori Ota, added an important piece to the puzzle when, working on a separate line of inquiry, found that in cultured cells and later-stage, post-implantation embryos the expression of Tead4 relied on a sector factor, Yap. This prompted Nishioka to look for a possible role for Yap in the early embryo as well. Looking at the distribution of the Yap protein, they were interested to find that while Yap localizes to the nucleus in outer cells, it is cytoplasmic in the inner ones. Importantly, this nuclear localization in outer cells precedes the expression of Cdx2, which suggested that, in these cells, Yap might trigger Tead4, which then switches on Cdx2. Overexpression of Yap or activated Tead4 caused Cdx2 to be expressed aberrantly in inner cells, while the expression of a Yap protein engineered to lack its Tead-binding domain had no effect. The question remained: What causes Yap to move to the nucleus only in outer cells? An experiment showed that in cultured cells a Hippo pathway member, Lats, phosphorylates Yap, causing it to remain in the cytoplasm. In the embryo, they found that phosphorylated Yap is only found in the inner cells, and that overexpression of Lats throughout the entire embryo causes a marked reduction of nuclear Yap and failure to form trophectoderm. Thinking about what might account for the inner cell-specific activity of Lats, Ota showed that the activation of Lats causes Yap to stay in the cytoplasm. Armed with this knowledge of what happens in vitro, Nishioka and colleagues did a series of experiments in 8-cell embryos in which he interfered with cell-cell adhesion, or dissociated and rearranged the cells of the embryo and found that cellular adhesion indeed does activate Hippo signaling, and that the adhesive environment determined by a cells position affects the localization of Yap within the cell. Other recent work from the Sasaki team had shown a role for Hippo signaling in the regulation of Tead in a phenomenon known as contact inhibition of proliferation. In this latest work, they have demonstrated a similar link between cell contact and differentiation in the early mammalian embryo, the first evidence of how a cells positional information determines its fate in this context. These new findings are an important step towards a better understanding of the mechanisms underlying differentiation along the inner-outer axis of the early embryo, says Sasaki. But there have also been reports that cell polarity is important in pre-implantation differentiation, and there is much we still do not know, so were looking forward to learning more about how such events are controlled during these critical early stages of development. |
|||||
|
|||||
 |
| Copyright (C) CENTER FOR DEVELOPMENTAL BIOLOGY All rights reserved. |