| RIKEN Center for Developmental Biology (CDB) 2-2-3 Minatojima minamimachi, Chuo-ku, Kobe 650-0047, Japan |
December 18, 2010 Cloning of animals by nuclear transfer remains an art as much as it is a science. Mouse cloning efficiency has lingered in the low single digits since the first success in 1998, at which time only 1-2% of all attempts was capable of generating cloned offspring. In the intervening years, that number has only risen by a few percentage points, and even when using the best available techniques, a cloning success rate of above 5% is considered high. A number of recent advances in cloning technology have involved the use of chemicals that interfere with the action of epigenetic regulators known as histone deacetylases (HDACs). But the reason why these inhibitors promote cloning success is unknown.
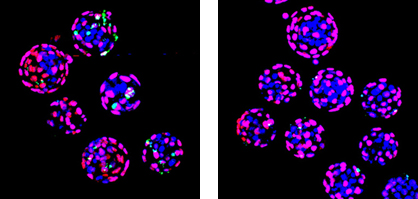
Tetsuo Ono and others in the Laboratory for Genomic Reprogramming (Teruhiko Wakayama, Team Leader) have now identified a pair of new HDAC inhibitors that improve the rate of full-term development in cloned mice. These new cloning boosters share a subset of characteristics with previously identified molecules of similar function, which may point the way to a better mechanistic understanding of how these new ingredients work. There are multiple types of HDAC, which are categorized into five classes (I, IIa, IIb, III and IV), and are expressed nearly ubiquitously, functioning as regulators of gene expression. Their inhibitors play an equally important role in fine-tuning their regulatory effects, but can also have cytotoxic effects, suggesting that the selection of inhibitor can have a major effect on whether it helps or harms the cloning process. To determine which works best in raising the efficiency of cloning and genomic reprogramming, Ono tested a number of HDACi proteins, with specificities for different classes of HDAC. These included well-established HDAC inhibitors, such as trichostatin A and scriptaid, as well as two new molecules, suberoylanilide hydroxamic acid (SAHA) and oxamflatin all of which inhibit HDAC classes I, IIa and IIb. The effects of valproic acid, an inhibitor of HDAC I and IIa, but importantly not of IIb were also examined. The team treated cloned embryos with varying concentrations of these HDAC and followed their development through hallmark stages such as blastocyst formation and full-term development. They found that at all stages, the embryos treated with trichostatin or the new factors SAHA and oxamflatin outperformed those treated with valproic acid and untreated controls. The new factors yielded particularly string improvements in cloning efficiency, with 9.4% of SAHA treated embryos and up to 7.5% of the oxamflatin-treated group reaching full term. Immunostaining of the cloned blastocysts revealed that the SAHA and oxamflatin-treated embryos had significantly higher numbers of cells in the inner cell mass (the population of cells that gives rise to the embryo proper), suggesting that these HAD inhibitors might enhance the reprogramming of somatic cell nuclei. TUNEL assays, which reveal apoptotic activity, showed that the SAHA and oxamflatin blastocysts had fewer cells undergoing programmed cell death, while those treated with valproic acid were more apoptotic. As a final functional test, Ono et al. generated embryonic stem cell lines from the cloned blastocysts (ntES lines) treated with the various factors, and found that HDACi-treated clones were two to three time more likely to be able to give rise to a pluripotent stem cell line. We wanted to know why only cloned embryos require HDACi treatment to encourage genomic reprogramming? says Wakayama. What we found was that perhaps the oocyte cytoplasm reprograms the somatic cell nucleus too strongly. By inhibiting a particular oocyte HDAC, we could achieve more appropriate reprogramming of donor nuclei, resulting in a higher cloning success rate.
|
|||||
|
|||||
 |
| Copyright (C) CENTER FOR DEVELOPMENTAL BIOLOGY All rights reserved. |