| RIKEN Center for Developmental Biology (CDB) 2-2-3 Minatojima minamimachi, Chuo-ku, Kobe 650-0047, Japan |
Jan 9, 2013 Insulin and insulin-like growth factors (IGFs) are known to play important roles in growth and metabolism. These hormones, which are conserved from insects to mammals, circulate in the blood and bind with specific cell surface receptors, triggering downstream signals within the cell. Although Insulin and IGFs are essential for regulating growth and metabolism but how these secreted molecules are regulated in the circulating blood has remained something of a puzzle. Naoki Okamoto and colleagues in the Lab for Growth Control Signaling (Takashi Nishimura, team Leader) have fitted a major new piece into this jigsaw, showing how a secreted decoy insulin receptor acts as a negative regulator of insulin/IGF signaling in Drosophila, thus controlling growth. This work is published in Genes & Development.
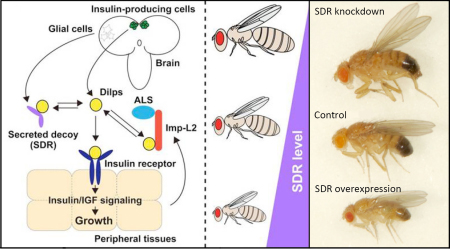
In vertebrates, secreted IGFs associate with IGF-binding proteins (IGFBPs) in the bloodstream, which stabilizes the IGFs and prevents them from binding with cell surface receptors. In the fly, Drosophila insulin-like peptides (Dilps) play the roles of both insulin and the IGFs in vertebrates. Previous reports have shown that the IGFBP-like protein Imp-L2 binds directly to several Dilps in the hemolymph, thereby regulating insulin/IGF signaling. But despite the need to keep such a crucial pathway under tight control, no other molecules capable of regulating Dilps have been reported in Drosophila. The Nishimura lab set out to identify novel molecules involved in developmental growth regulation by screening secreted protein RNAi knockdown phenotypes for effects on body size, and identified a gene whose loss of function resulted in a higher growth rate in larval stages and larger adult body sizes. This new gene included a sequence closely similar to that of the insulin receptor (InR) extracellular domain, which, along with its functional attributes, led them to name it secreted decoy of InR (SDR). SDR mutants showed no defects in survival or fertility, but grew about 20% larger than wildtype flies. Close observation of wings in adult mutants revealed that the change in overall size was a result both of greater numbers of cells and increased cell size. SDR has neither a transmembrane nor a cytoplasmic domain, pegging it as a likely secreted protein, which Okamoto et al confirmed by detecting high levels of the factor in hemolymph. The team next looked at gene expression patterns and conditional RNAi phenotypes in various tissues and found that it was highly expressed in the glial cells of the brain. After deciphering where SDR was made, the next step was to find out how it worked. Using the localization of dFOXO, which responds to insulin/IGF signaling on/off cues, as an index, they found that insulin/IGF signaling was accelerated in SDR mutants, and conversely inhibited on its overexpression. In vitro assays further showed direct binding between SDR and several Dilps, suggesting that its mode of action involves antagonistic binding with these IGFs, preventing their association with receptors. Other insulin-linked molecules are known to respond to changes in nutritional status. Looking at starved flies, in which the expression of some Dilps is down-regulated and that of Imp-L2 increased, a mechanism though to allow the body to sacrifice growth in favor of storing nutrients, they found that SDR expression and secretion remained unexpectedly stable under low-nutrition conditions. But when SDR mutants were bred under extremely adverse conditions where food availability was only one-tenth normal, the failure of growth inhibition led to poor survival, with over half of the flies dying during metamorphosis, suggesting that SDR is an essential safety mechanism for controlling insulin/IGF signaling under poor nutritional conditions. Although the SDR gene is found only in insects, similar decoy systems for Insulin/IGF signaling are likely present in other species. Recent observations have indicated that mammalian insulin receptor can potentially produce a soluble decoy, says Nishimura. Our initial focus on was on the role of SDR in growth regulation, but from now we will be interested to learn more about its functions in other phenomena, such as metabolism and aging. |
|||||
|
|||||
 |
| Copyright (C) CENTER FOR DEVELOPMENTAL BIOLOGY All rights reserved. |