| RIKEN Center for Developmental Biology (CDB) 2-2-3 Minatojima minamimachi, Chuo-ku, Kobe 650-0047, Japan |
February 1, 2013 Networks of tubular structures form the basis for many important organs and tissues in the animal body, such as the vasculature and airways. The need for such luminal structures developed as bodies grew larger over the course of evolution, necessitating conduits for the life-sustaining circulation of nutrients, gases and wastes throughout the body. Animals and their organs come in many different sizes and shapes, of course, so it is also necessary for lumens to fit the surrounding morphology. Width and length are the critical parameters in this equation, but how does the developing embryo make these measurements? New work by Bo Dong and others in the Lab for Morphogenetic Signaling (Shigeo Hayashi, Group Director) now shows how an intracellular mechanism known as retrograde trafficking establishes the length of the developing trachea in fruit fly embryos. Published in Nature Communications, this study sheds light on the morphological regulation of an important class of tissue structures.
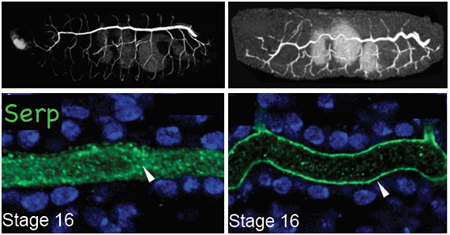
The formation of the Drosophila trachea is under the control of extracellular matrix proteins such as chitin secreted within its lumen. It has previously been shown that the diameter of this structure is regulated by an intracellular protein trafficking pathway mediated by actin complexes, while its length is controlled by a chitin deacetylase known as Serpentine (Serp). Loss of Serp results in an abnormally long trachea, but just how this mechanism is tied to the protein trafficking that regulates intraluminal secretion was poorly understood. After searching for proteins showing specific localization patterns in the cells of the trachea, Dong et al. came to focus on Rab9, a protein known to be involved in retrograde trafficking, a kind of intracellular recycling process in which secreted molecules in the cells environment are endocytosed and carried in endosomes to the Trans-Golgi network, where they are modified and readied to be secreted again. Generating a rab9 mutant, the group noted that while the width of the trachea and embryogenesis as a whole appeared normal, the trachea grew to unusual length, a phenotype resembling that seen in Serp mutants. Looking for co-localization of the two molecules, they found that after about 12 hours of development, the levels of Serp expression within the tracheal lumen dropped sharply, while chitin and other proteins were unaffected. In mutants of another known retrograde trafficking protein, Vps35, the over-long trachea phenotype was also observed, and biochemistry assays indicated that Rab9 and Vps35 bind each other directly. Using a live imaging system, Dong watched the dynamics of these three proteins and found that they co-localize as assemblies in an endosomal subtype known as late endosomes. Knowing that actin is crucial for the budding of vesicles from endosomes, they next looked for a spatial relationship between actin and the Rab9 endosomes. They found that actin and a newly identified actin-associated factor, WASH, localized with Vsp35 at budding sites and are involved in the endosomal recycling of Serp. We think it is very interesting that loss of retrograde trafficking does not affect the tracheal diameter, which suggests that length and width are under the control of separate trafficking pathways, says Hayashi. Retrograde trafficking is widely conserved in animals, including humans, so we will be eager to discover whether it plays a more general role in morphogenesis. |
|||||
|
|||||
 |
| Copyright (C) CENTER FOR DEVELOPMENTAL BIOLOGY All rights reserved. |